|
When you’re measuring extremely tiny amounts of a substance, it can be helpful to use molar mass. One mnemonic device for remembering diatomic elements (molecules of 2 atoms) is: Have No Fear Of Ice Cold Beverages (Hydrogen, Nitrogen, Fluorine, Oxygen, Iodine, Chlorine, Bromine).Multiply them by the molar mass constant, and then multiply the result by 2. This means that if you want to find the molar mass of elements that are composed of 2 atoms, such as hydrogen, oxygen, and chlorine, then you'll have to find their relative atomic masses. Some elements are only found in molecules of 2 atoms or more.This converts atomic units to grams per mole, making the molar mass of hydrogen 1.007 grams per mole, of carbon 12.0107 grams per mole, of oxygen 15.9994 grams per mole, and of chlorine 35.453 grams per mole. This is defined as 0.001 kilogram per mole, or 1 gram per mole. 
The molar mass of all these above compounds is calculated same as we calculate the H2O molar mass and other compounds.Multiply the relative atomic mass by the molar mass constant. Calculate the molar mass of Calcium hydroxide?.Calculate the molar mass of Oxalic acid?.Calculate the molar mass of Hydrosulfuric acid?.Molar mass of CH 3CH 2OH = 46.08 g/mol Test Yourself Molar mass of CH 3CH 2OH = (Atomic mass of C) + 3 ( Atomic mass of H) × (Atomic mass of C) + 2 ( Atomic mass of H) × (Atomic mass of O) + ( Atomic mass of H) Now we calculate the molar mass of ethanol: The chemical formula of this compound is CH 3CH 2OH. Molar mass of C 6H 12O 6 =180.18 g/mol How do you find the molar mass of ethanol? Molar mass of C 6H 12O 6 = 6 (Atomic mass of C) + 12 ( Atomic mass of H) + 6 (Atomic mass of O) Molar mass of NH 3 = 17.04 g/mol What is the molar mass in g mol for glucose sugar C 6H 12O 6? Molar mass of NH 3 = (Atomic mass of N) + 3 (Atomic mass of H) Look at the atomic masses of nitrogen and hydrogen. It contains nitrogen and hydrogen elements. 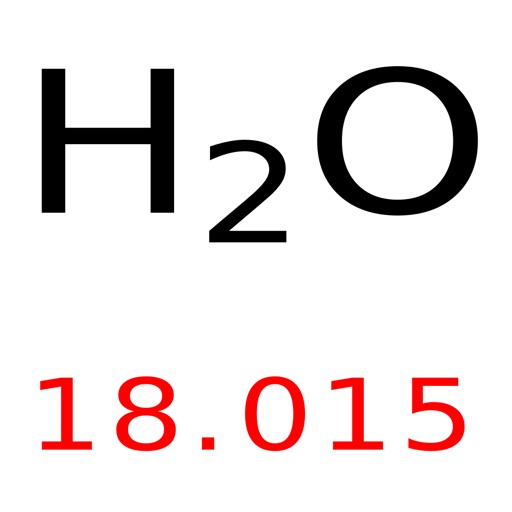
The molecular geometry of NH 3 is trigonal pyramidal. NH 3 is the molecular formula of ammonia. Molar mass of H 2SO 4 = 98.08 g/mol How do you find the molar mass of NH3? Molar mass of H 2SO 4 = 2 (Atomic mass of H) + (Atomic mass of S) + 4 (Atomic mass of O) The atomic masses of these elements are given below: Sulfuric acid contains hydrogen, sulfur, and oxygen elements.  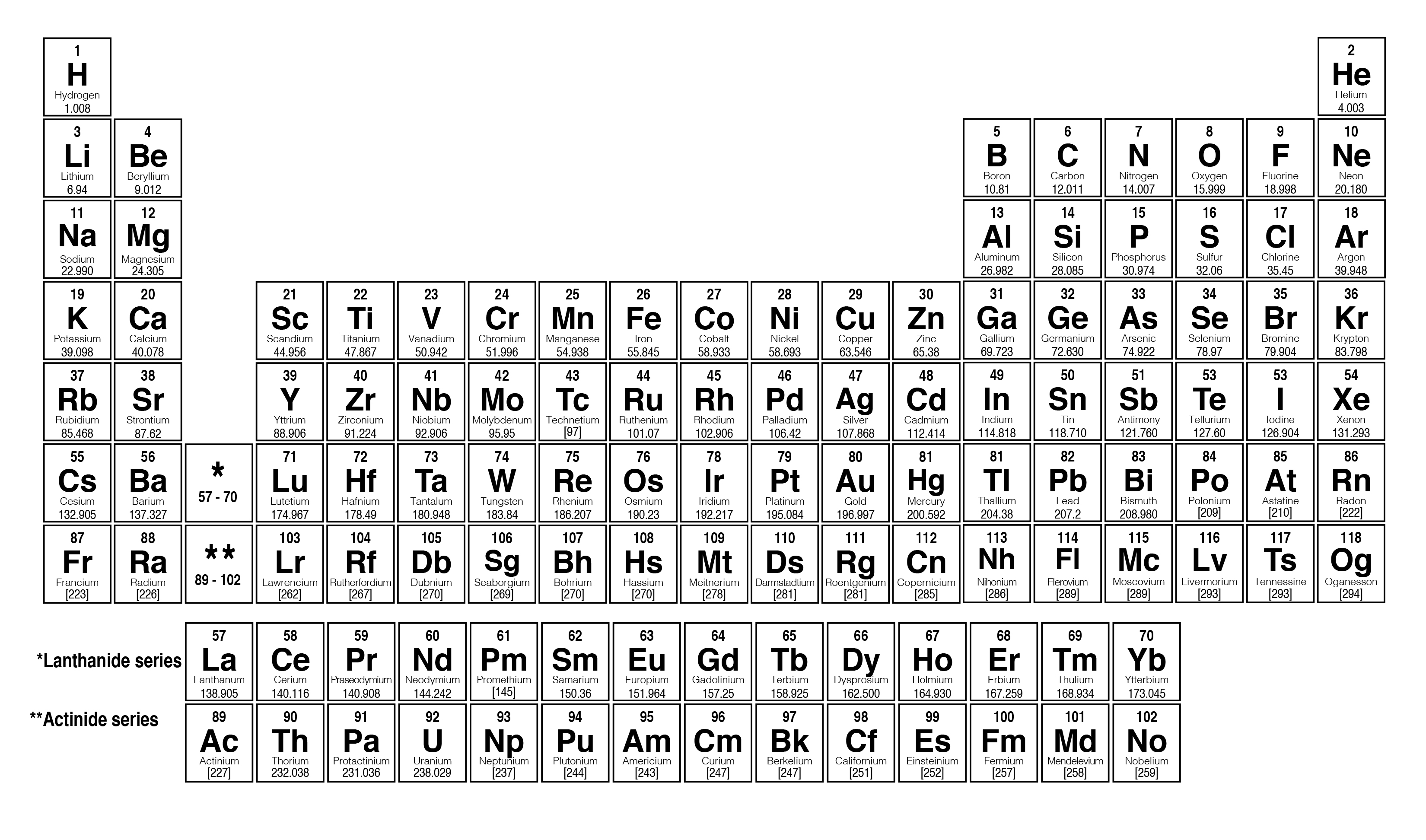
The molar mass of sulfuric acid is 98.08 g/mol. H2O molar mass = 18.02 g/mol How to calculate the molar mass step by step? What is the molar mass of H 2SO 4? H2O molar mass = 2 (Atomic mass of H) + (Atomic mass of O) In this way, we calculate the molar mass of water. One thing kept in mind is that in a water molecule, there are two hydrogen atoms present so we multiply the atomic mass of hydrogen by 2 and then add it with oxygen atomic mass. So we go to the periodic table and find the atomic masses of oxygen and hydrogen. Water molecules are made up of one oxygen atom and two hydrogen atoms. Let’s Find Out the H2O Molar Mass?īefore calculating the H2O molar mass, we need to know the atoms that make water. For example at the top right corner, hydrogen is present and the atomic mass of hydrogen is given below the symbol of hydrogen which is 1.01. The atomic masses of each element are given right below the symbol of an element. There are many names for molar mass and it tells us the mass of one mole of a given compound. How to calculate the H2O molar mass? Here we discussed the easiest method to calculate the molar mass of different compounds.
0 Comments
Leave a Reply. |
Details
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
